AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Stem cell therapy for knees9/13/2023  
However, current clinical studies have limited evidence regarding study objectives, test designs, and patient populations. SRs/MAs results revealed that stem cell therapy relieved pain in patients over time but did not improve knee function. 
However, these two studies suggest that stem cell therapy does not bring significant ARs to patients. Additionally, two studies have reported cases of prostate cancer and breast tumors, respectively. Some studies reported over 10% prevalence of knee pain (24.5% 95% CI ), knee effusion (12.5% 95% CI ), and knee swelling (11.9% 95% CI ). Nineteen ARs were reported in 50 studies, including five knee-related ARs, seven common ARs, and seven other ARs. Resultsįifty clinical studies and 13 systematic reviews/meta-analyses (SRs/MAs) were included. Two researchers conducted literature screening and data extraction independently, and the evidence quality was evaluated according to the Institute of Health Economics and AMSTAR 2 criteria. Quantitative systematic reviews of stem cell therapy for KOA that conducted meta-analysis were included. Clinical studies that reported adverse reactions (ARs) of stem cell therapy in KOA patients were included without limiting the type of studies. Using “stem cells” and “knee osteoarthritis” as the search terms, several databases, including PubMed, Web of Science, Cochrane, Embase, and, were searched on August 25, 2022, and updated on February 27, 2023. A comprehensive evaluation of the safety and efficacy of stem cell therapies and scientific evidence quality is necessary. However, it remains uncertain whether the current scientific evidence supports the clinical application of stem cells in treating KOA. If you are interested in cell therapy, the best course of action is to speak with your physician, as these treatments need to be evaluated for safety and effectiveness in clinical trials approved by Health Canada.The success of stem cell therapy for knee osteoarthritis (KOA) in preclinical animal models has accelerated the pace of clinical translation. 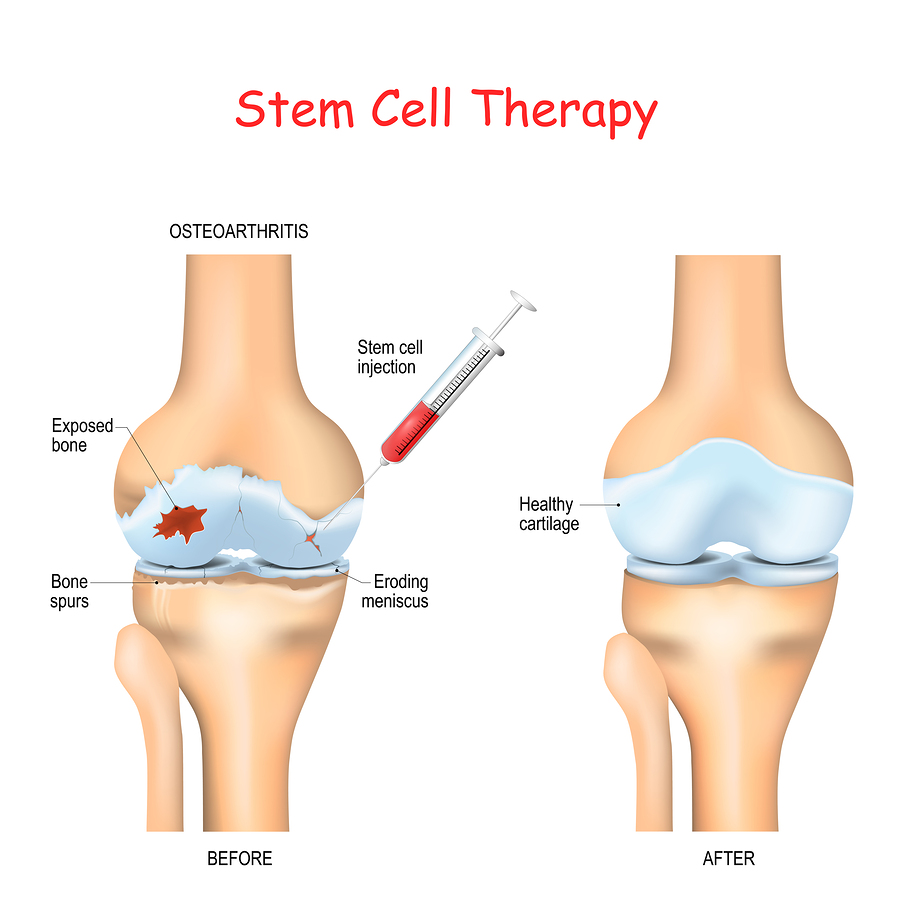
The Arthritis Society funds research on cell therapy in arthritis to further our understanding of the short- and long-term effectiveness and safety of these treatments in managing or preventing conditions like arthritis. Cell therapy research is increasingly becoming more common and sophisticated, which may lead to more effective tissue repair and regeneration techniques for arthritis and other conditions in the future. With further research, this may have the potential to reverse the damage of arthritis and avoid the need for joint replacement surgery. Cell therapy research (which can include but is not limited to stem cells) offers the promise for a person’s own cells to potentially be used to repair or replace cells damaged by disease or injury or reduce inflammation and pain. The Arthritis Society supports research on cell therapy and ‘stem cells’, which are studied under regulated clinical trials. These treatments often do not help and may even harm patients. However, hype about stem cell therapy has led some people to pay thousands of dollars on treatments that promise to cure ailments from arthritis to Parkinson’s disease. This allows them to multiply and repair damage. You may be familiar with ‘stem cells’, which can renew themselves and mature into specialized cells like cartilage, heart or brain cells. Health Canada does not allow clinics and providers in Canada to offer cell therapy These are not approved by Health Canada, which considers all ‘stem cell’ treatments as drugs and requires them to be proven in clinical trials. However, there are some for-profit clinics which offer cell or even ‘stem cell’ treatments for osteoarthritis and other musculoskeletal diseases. Many cell therapies for arthritis – those that use a patient’s own cells – are still experimental and are being evaluated in regulated clinical trials.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
